How Do These Drugs Work?
Lyrica is the brand name for the drug pregabalin, and gabapentin is the name of the medication that is used in drugs such as Neurontin, Gralise, and Horizant. All of these drugs are prescribed as anticonvulsant or antiepileptic medications. Pregabalin and gabapentin are structurally similar to the brain chemical (neurotransmitter) gamma-aminobutyric acid (GABA), which is the major inhibitory neurotransmitter in the central nervous system.1
The exact way that Lyrica and gabapentin work is not well understood, but they do not appear to act on GABA neuronal systems, as one would expect given their chemical structure. Instead, they may bind to voltage-gated calcium channels in the central nervous system (CNS), which may decrease neurotransmitter release throughout the CNS.1
Differences Between Lyrica (Pregabalin) and Gabapentin
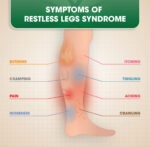
In the U.S., medicines containing gabapentin are approved to treat seizures, post-herpetic neuralgia, and restless legs syndrome. Gabapentin also has numerous off-label uses, including the management of neuropathic pain, insomnia, anxiety, bipolar disorder, menopausal conditions (i.e., hot flashes), vertigo, itching of the skin, and migraines.
A major difference between Lyrica and gabapentin is that the DEA has classified Lyrica as a controlled substance.
Lyrica is also approved to treat seizures and post-herpetic neuralgia. In addition, it can treat neuropathic (nerve) pain associated with diabetes (i.e., diabetic peripheral neuropathy) and pain associated with fibromyalgia.
A major difference between Lyrica and gabapentin is that the Drug Enforcement Administration (DEA) has classified Lyrica as a controlled substance (listed in Schedule V), whereas medicines containing gabapentin are not currently scheduled as controlled substances. This means that Lyrica and substances containing pregabalin are considered to have some potential for abuse and physiological dependence.
Despite not being a scheduled, controlled substance, there are documented cases of gabapentin abuse. A review of cases of gabapentin misuse found that the main reasons people abused the drug were to get high, ease opioid withdrawal symptoms, and increase the effects of methadone. Another study found that the rates of abuse ranged from 40% to 65% among people with a prescription for the drug and 15% to 22% among populations that abuse opioids. Many people who abused the drug had a history of substance abuse.
A review of the 2 drugs concluded that “pregabalin appears to have some distinct pharmacokinetic advantages over gabapentin.” Lyrica appears to be absorbed more rapidly than gabapentin, with peak blood plasma concentrations occurring within 1 hour. The amount of the drug in a person’s system (bioavailability) also remains the same regardless of the dose. In contrast, gabapentin’s bioavailability decreases from 60% to 33% when the dose is increased from 900mg to 3600mg per day.
In addition, Lyrica may be a relatively more potent anticonvulsant and neuroleptic medication—leading to a greater reduction in seizure frequency in patients and being more effective for treating neuropathic pain.
Finally, both drugs can lead to withdrawal if a person suddenly stops using them or reduces the dose. Abrupt cessation can lead to status epilepticus in patients (seizures that don’t stop).6
Gabapentin withdrawal symptoms include:
- Drowsiness.
- Lack of control over muscle movements.
- Fatigue.
- Nausea.
- Vomiting.
- Dizziness.
- Anxiety.
- Insomnia.
- Sweating.
Pregabalin withdrawal symptoms include:
- Headaches.
- Nausea.
- Diarrhea.
- Sleep problems.
- Sweating.
- Anxiety.
Experts recommend that people who are taking antiepileptic drugs taper off them gradually to reduce the risk of withdrawal symptoms and increased seizures. Lyrica may be gradually discontinued over the course of at least one week, but only your prescribing physician can best determine the right timeframe/tapering schedule for you.

If you have been misusing either or both of these medications, you could be at risk of a potentially complicated withdrawal, and should seek the help of a medical detox program or other medical professional guidance.
Side Effect Potential
Though the side effect profiles of both drugs are similar, some studies have found that Lyrica may have somewhat fewer of them. Dizziness and sleepiness are the most common side effects associated with both drugs, but more serious concerns such as suicidal ideation have also been reported.
Side effects of gabapentin include:
- Drowsiness.
- Dizziness.
- Shaking.
- Headache.
- Blurred vision.
- Memory problems.
- Dry mouth.
- Constipation.
- Increased appetite.
- Weight gain.
- Back or joint pain.
- Fever.
- Ear pain.
- Runny nose.
- Flu-like symptoms.
- Red, itchy eyes.
- Poor coordination.
- Shortness of breath.
- Itching.
- Swelling of the face, tongue, lips, or eyes.
- Rash.
- Seizures.
Side effects for Lyrica include:
- Headache.
- Dry mouth.
- Blurred vision.
- Constipation.
- Increased appetite.
- Weight gain.
- Back pain.
- Flu-like symptoms.
- Red, itchy eyes.
- Gas and bloating.
- Poor coordination.
- Shortness of breath.
- Wheezing.
- Chest pain.
- Rash.
- Memory problems.
The Bottom Line
Gabapentin and Lyrica are similar in their chemical composition and in the conditions they are approved to treat. However, some research studies suggest that Lyrica has some advantages, particularly with the control of certain types of pain.
Based on the current classification by the DEA, pregabalin is considered to have a higher risk for abuse and for the development of physical dependence than products containing gabapentin. The general rule is that both drugs be discontinued on a tapering schedule (lowering the dose at specific intervals) to avoid potential withdrawal symptoms even in individuals who use these drugs for medical reasons.
The decision to use products containing gabapentin or pregabalin should be made during a consult with one’s physician. A doctor will take into account your medical history, symptoms, age, and other factors before writing a prescription. If you are prescribed either drug, always take it according to your doctor’s instructions and never share your prescription with other people.
Fear and anxiety for legitimate users of pregabalin and gabapentin
Home Office proposals to reclassify both gabapentin and pregabalin – commonly used to treat neuropathic or nerve pain – as class ‘C’ substances, will make access to them more difficult. Publicity surrounding the proposal has also created fear and anxiety and left people who depend on gabapentin or pregabalin for pain relief, questioning the safety of the two medicines which, if used responsibly and with the right combination of support, can change the lives of people who have previously struggled to manage pain symptoms.
Heather Wallace, Chair of Pain Concern – a UK charity based in Scotland – acknowledges the rise in the number of deaths associated with pregabalin and gabapentin but says the circumstances around the alarming statistics haven’t been properly examined.
Heather said: “We have known for a long time that these medicines have been over-prescribed, but the people who find them beneficial for pain relief, haven’t misused them and feel they are being punished for something they haven’t done. In many cases it is difficult to find the right combination of pain relief, and we know from our helpline that people have been left fearful and confused by this proposal and all the publicity it has generated. It’s premature and risks harming people who are using these medicines because there is nothing else for them that works.”
Pain specialist Dr Pamela Bell believes that open and informed discussion between people living with pain and their GP about the benefits versus the risks of potential treatment, including the risks of dependency, addiction and interactions with other drugs, is vital. “We know many people who benefit from these medicines when they are appropriately prescribed and we share their concerns at moves to reclassify pregabalin and gabapentin. Many people tell us it has taken years for them to find the right combination of medication, physiotherapy and support that allows them a better quality of life and it will prove more difficult to access pregabalin and gabapentin when they are reclassified as controlled drugs.”
Pain Concern works to empower people living with long term pain to be proactive in managing their condition and has in response to the demand for trustworthy and reliable information on pain medication has produced three information leaflets on: gabapentin and pregabalin; amitriptyline and opioids. Written by pain experts, the leaflets provide up-to-date information and facts to help people discuss possible treatments and therapies with their GP or clinic.
How Gabapentin Differs From Pregabalin ?
Neuropathic pain causes significant morbidity in the United States.
The incidence of peripheral neuropathy is estimated at about 2.4% of the population.1 Of the 14 million US individuals with diabetes, roughly 25% experience painful diabetic neuropathy.
Despite advances in vaccination for varicella zoster virus, around 25% of patients with a herpes zoster infection will develop persistent neuropathic pain.2 More than 85% of patients with neuropathic pain caused by peripheral neuropathy will require pharmacotherapy.
Unfortunately, there are few head-to-head trials comparing agents for neuropathic pain, so selecting the best option can be difficult.
Pregabalin and gabapentin are often considered first-line treatments for various neuropathic pain syndromes, generally irrespective of cause. Because the products are so variable, this article compares the pharmacokinetics (PK) and pharmacodynamics (PD) of pregabalin with various gabapentin formulations, and also covers conversion regimens.
Pharmacokinetics of pregabalin and gabapentin
Both pregabalin and gabapentin are antiepileptic medications that bare structural resemblance to gamma-aminobutyric acid (GABA), though neither agent has activity in GABA’s neuronal systems.
Although the exact mechanism of action is somewhat unclear, the drugs’ efficacy in neuropathic pain is linked to their ability to bind to voltage-gated calcium channels in the central nervous system (CNS), specifically to the alpha-2-delta protein. This binding decreases neurotransmitter release in the CNS as a result of reduced calcium influx through the gated channels. Gabapentin is indicated as adjunct therapy for partial seizures and postherpetic neuralgia. Pregabalin is indicated for the same uses as gabapentin, plus the management of fibromyalgia and neuropathic pain associated with diabetes, specifically diabetic neuropathy.
Overall, the pharmacokinetic profiles of these 2 medications are somewhat similar, but they also have some significant differences.
For example, both drugs are structurally similar to the amino acid leucine. Because of this, they can both undergo facilitated transport across cellular membranes through system L-amino acid transporters.3 This is the major form of absorption for gabapentin and pregabalin, with the exception of an extended-release gabapentin prodrug to be discussed later.
However, pregabalin may either have an additional system of absorption or be better transported than gabapentin, as it is almost completely absorbed, while gabapentin is not. In addition, absorption of gabapentin is limited to the small intestine, while pregabalin is absorbed throughout the small intestine and extending to the ascending colon.
Gabapentin is more slowly and variably absorbed, with peak plasma concentrations around 3 hours post-dose. Pregabalin is quickly absorbed, with the maximum rate of absorption being 3 times that of gabapentin. It reaches peak blood concentrations within an hour after ingestion.
Absorption of gabapentin is saturable, leading to a non-linear pharmacokinetic profile. As gabapentin doses increase, the area under the curve (AUC) does not follow proportionally. Unlike gabapentin, absorption of pregabalin is not saturable, and the drug has a linear pharmacokinetic profile.
The bioavailability of generic gabapentin in tablet and capsule formulations equivalent to brand-name Neurontin is about 80% at lower doses such as 100 mg every 8 hours, but only 27% bioavailable at doses of 1600 mg every 8 hours.3,4 This differs greatly from pregabalin, which boasts a greater than 90% bioavailability across a dosage range from 75 mg to 900 mg daily in divided doses.
Gabapentin’s bioavailability for its intended patient population is also more variable than pregabalin’s bioavailability. Variability among patients is believed to be 10% to 15% with pregabalin and 20% to 30% with gabapentin.
Finally, food increases the AUC of gabapentin by about 10%, with no change in time to maximum concentration (tmax). In contrast, the AUC of pregabalin is unaffected by food, though absorption is slower.
Distribution of gabapentin and pregabalin is very similar. Neither agent is bound by great extent to any plasma proteins, decreasing the likelihood of drug interactions due to protein binding. Both have high aqueous solubility, and the volume of distribution of each is similar (0.8 L/kg and 0.5 L/kg for gabapentin and pregabalin, respectively).
Drug-drug interactions are unlikely for both pregabalin and gabapentin. Neither pregabalin nor gabapentin is affected by cytochrome (CYP) drug interactions, as neither drug is metabolized by CYP enzymes. Both undergo metabolism to a negligible extent (<1%).
Renal excretion is the major method of both drugs’ elimination from the body. Agents that decrease small bowel motility can theoretically cause an increase in the absorption of gabapentin, because it is not completely absorbed. However, as pregabalin is more than 90% absorbed, its absorption is not affected by changes in small bowel motility.
Gabapentin formulations
Gabapentin is also available in 2 extended-release formulations: a tablet (Gralise) and a gastro-retentive prodrug, gabapentin enacarbil (Horizant).
Both have different pharmacokinetics and are not interchangeable with standard formulations, the original of which is Neurontin. Side effects of these newer formulations are similar to standard formulations.
Gralise is indicated for postherpetic neuralgia and taken as an 1800 mg maintenance dose once a day. The AUC of 1800 mg of Gralise is slightly less than 1800 mg of the standard formulation. In addition, the average maximum concentration (Cmax) of Gralise is slightly higher than 1800 mg of the standard form, and minimum concentration (Cmin) is slightly lower. Finally, Tmax is increased in the extended-release form.
Although food does not palpably affect the AUC of standard forms of gabapentin, Gralise should be taken with food because the bioavailability is greatly increased (33% to 118%, depending on the meal’s fat content).
Horizant is a prodrug of gabapentin indicated for postherpetic neuralgia and restless leg syndrome. The recommended maintenance dose is 600 mg twice a day. Doses greater than 1200 mg daily are not recommended, as side effects increase without a corresponding increase in efficacy.7
Bioavailability of gabapentin enacarbil is about 75%, which is somewhat improved over the standard formulation. It is absorbed through the small intestine through a proton-linked monocarboxylate transporter (MCT-1).
Unlike the original formulation, absorption of gabapentin enacarbil is not saturated at high doses, as MCT-1 is expressed in high levels in the intestinal tract. The drug undergoes near-complete first pass hydrolysis to gabapentin by non-specific carboxylesterases mainly in enterocytes, as well as in the liver to a lessor degree.
Consumption of alcohol increases the release of gabapentin enacarbil from the extended-release tablet. Therefore, alcohol should be avoided when taking Horizant due to increased risk of side effects.
Like standard formulations of gabapentin, Gralise and Horizant are not metabolized to an appreciable extent by phase I metabolism, and they are neither a substrate nor an inhibitor of p-glycoprotein. All forms of gabapentin must be adjusted in renal dysfunction, similar to standard formulations.
One review found that these extended-release formulations have similar efficacy to standard ones, and they might also have fewer adverse events.
Both pregabalin and gabapentin are well tolerated. Dizziness and somnolence are the most common side effects in both drugs (>20% seen in gabapentin).3-5 Confusion and peripheral edema have also been reported with gabapentin.
With both drugs, side effects are dose dependent and reversible if the medication is discontinued.3 The abrupt discontinuation of any form of gabapentin is not recommended because withdrawal symptoms such as anxiety, insomnia, nausea, pain, and sweating may present. When discontinuing gabapentin it is recommended to taper the dose over a week at least.
As with all antiepileptic drugs, increased risk of suicidal ideation is possible.
Pharmacodynamics of pregabalin and gabapentin
Gabapentin and pregabalin vary in terms of binding affinity and potency. Pregabalin has an increased binding affinity for the alpha-2-delta protein and is a more potent analgesic in neuropathic pain compared with gabapentin.
One study developed a population pharmacokinetic model comparing pregabalin with gabapentin.9 The authors calculated values for the concentration of the drug that will give one-half the maximum pharmacologic response (EC50) and used these values to assess potency of the 2 medications.
Based on studies of gabapentin and pregabalin in epilepsy, the EC50 values of pregabalin and gabapentin were estimated to be about 9.77 mg/mL and 23.9 mg/mL, respectively.9 From these data, pregabalin was estimated to be about 2.4 times more potent. For neuropathic pain, pregabalin’s potency ratio may be even greater.
Using studies in postherpetic neuralgia, the EC50 values of pregabalin and gabapentin were estimated to be about 4.21 mg/mL and 11.7 mg/mL, respectively. Based on these values, pregabalin was estimated to be about 2.8 times more potent than gabapentin.
Pregabalin and gabapentin differ somewhat in terms of their dose-response curves.
One study analyzed data from phase 2 trials of gabapentin and pregabalin and created a pharmacodynamic model.3 The authors found that in patients with postherpetic neuralgia, mean pain scores decreased as the dose of both gabapentin and pregabalin increased.
However, the study also uncovered a plateau of gabapentin’s effect on reducing pain at around 3600 mg/day. In contrast, the pain-relieving effect of pregabalin continued to increase up to the maximum dose of 450 mg/day.
Pregabalin also exhibited a steeper dose-response curve than gabapentin. Based on the dose-response curves predicted in this model, a dose of 450 mg/day of pregabalin equates to about 3600 mg/day of gabapentin.
Converting from gabapentin to pregabalin
For clinicians who wish to convert patients from gabapentin to pregabalin, there are a few studies that reviewed such a conversion.
One cohort study reviewed the utility of switching patients with neuropathic pain due to peripheral neuropathy from gabapentin to pregabalin. The study followed patients who were switched from gabapentin to pregabalin and then compared them to those who stayed on gabapentin. The authors also stratified the pregabalin group further into those who responded well or poorly to gabapentin, with gabapentin stopped after the nighttime dose and pregabalin started the following morning.
Dosages were switched using the following algorithm:
- Gabapentin ≤900 mg/day → pregabalin 150mg/day
- Gabapentin 901 mg/day to 1500 mg/day → pregabalin 225 mg/day
- Gabapentin 1501 mg/day 2100 mg/day → pregabalin 300 mg/day
- Gabapentin 2101 mg/day 2700 mg/day → pregabalin 450 mg/day
- Gabapentin >2700 mg/day → pregabalin 600 mg/day
This rapid change was generally well tolerated by patients.
The authors found that those who responded well to gabapentin and those who did not showed additional benefit with decreased pain when they were switched to pregabalin. Patients taking pregabalin also had improved pain control compared with those who remained on gabapentin.
Switching to pregabalin resulted in improved pain relief and also fewer adverse events. This was particularly true for patients who previously responded to gabapentin.
Patients who experienced adverse events with gabapentin were more likely to also experience adverse events with pregabalin. These patients were also more likely to discontinue use of pregabalin than those who responded well to both gabapentin and pregabalin.
Another small trial compared the degree of pain relief with gabapentin to pregabalin in patients with postherpetic neuralgia in order to more closely determine equivalent dosing between the 2 medications.
Patients were switched from gabapentin to pregabalin using one-sixth the dose of gabapentin with unchanged dosage frequency. After switching medications, patients reported similar pain relief and side effects, with the exception of an increased incidence of peripheral edema in the pregabalin group.
The authors concluded that the analgesic effect of pregabalin was about 6 times that of gabapentin.
Other studies have looked at methods for converting gabapentin to pregabalin. One such trial used population pharmacokinetic models to examine 2 possible scenarios for converting gabapentin to pregabalin, using a ratio of 6:1 gabapentin to pregabalin.
The first scenario involved discontinuing gabapentin and immediately starting pregabalin at the next scheduled dosing period. The other option included a gradual transition from gabapentin to pregabalin.
In this second scenario, the gabapentin dose was decreased by 50%, and 50% of the desired pregabalin dose was given concurrently for 4 days. After this time, gabapentin was discontinued and pregabalin was increased to full desired dose.
The model looked at transitioning patients from gabapentin to pregabalin at various doses, including:
- Gabapentin 900 mg/day → pregabalin 150 mg/day
- Gabapentin 1800 mg/day → pregabalin 300 mg/day
- Gabapentin 3600 mg/day → pregabalin 600 mg/day
Both scenarios were quick and seamless, so the authors concluded that either technique could be an effective method to switch patients between the medications.9
Final thoughts
Though pregabalin and gabapentin have somewhat similar pharmacokinetic and pharmacodynamic profiles, there are clearly significant differences. Overall, pregabalin has more predictable pharmacokinetics, and it also shows a stronger binding affinity to its target receptor, increased potency, and a steeper dose-response curve in neuropathic pain that does not plateau over recommended dosing levels.
A few studies have found that pregabalin has fewer side effects and may be more efficacious for neuropathic pain than gabapentin. Several studies reviewing conversion of gabapentin to pregabalin predict that a rough ratio for conversion is about 6:1 gabapentin to pregabalin. In addition, a direct switch from gabapentin to pregabalin seems to be well tolerated, making the conversion simple.
Clinicians should note that pregabalin has various pharmacokinetic and pharmacodynamic advantages over gabapentin, and a conversion between the 2 medications is often well tolerated.