The gate control theory of pain, put forward by Ron Melzack and Patrick Wall in 1962, is the idea that physical pain is not a direct result of activation of pain receptor neurons, but rather its perception is modulated by interaction between different neurons.
The gate control theory of pain asserts that non-painful input closes the “gates” to painful input, which prevents pain sensation from traveling to the central nervous system. Therefore, stimulation by non-noxious input is able to suppress pain.
First proposed in 1965 by Ronald Melzack and Patrick Wall, the theory offers a physiological explanation for the previously observed effect of psychology on pain perception.
Combining early concepts derived from the specificity theory and the peripheral pattern theory, the gate control theory is considered to be one of the most influential theories of pain because it provided a neural basis which reconciled the specificity and pattern theories and ultimately revolutionized pain research.

Unlike an actual gate, which opens and closes to allow things to pass through, the “gate” in the spinal cord operates by differentiating between the types of fibers carrying pain signals. Pain signals traveling via small nerve fibers are allowed to pass through, while signals sent by large nerve fibers are blocked. Gate control theory is often used to explain phantom or chronic pain.
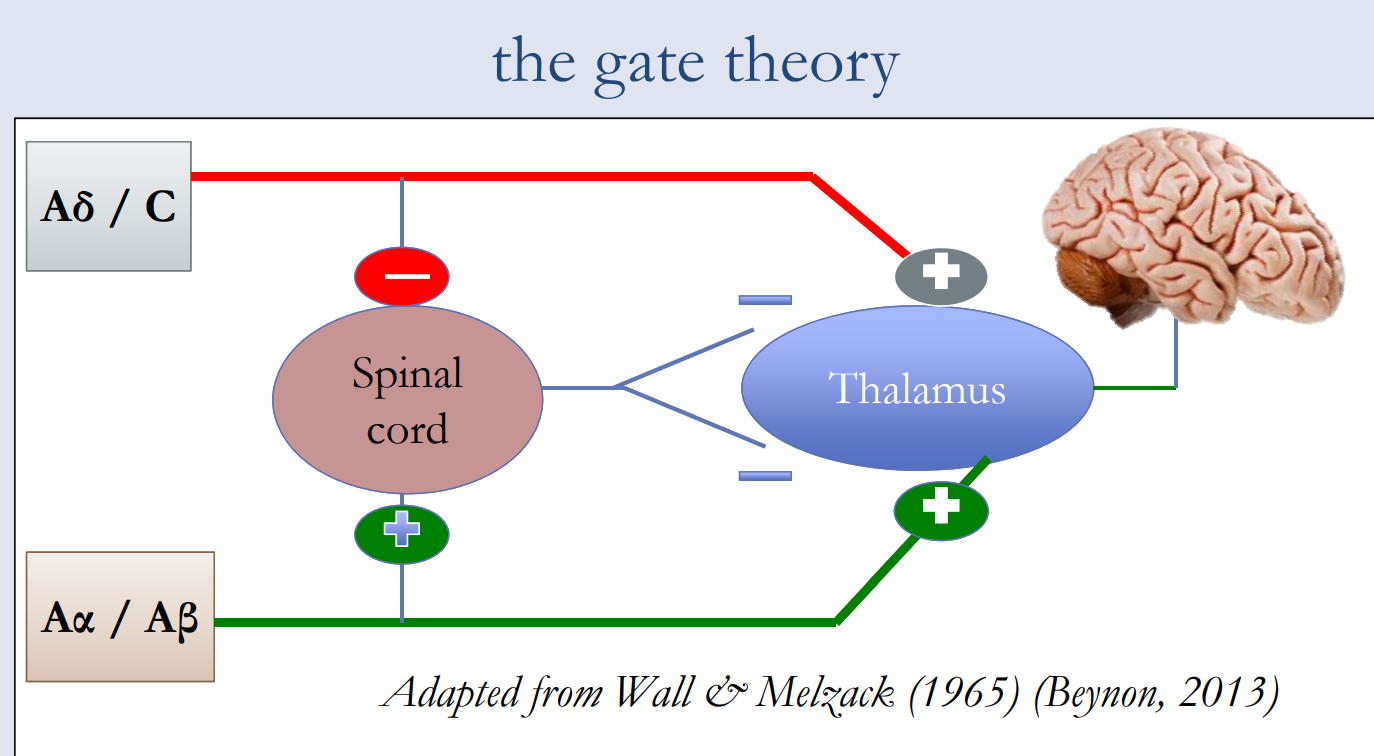
The interplay among these connections determines when painful stimuli go to the brain:
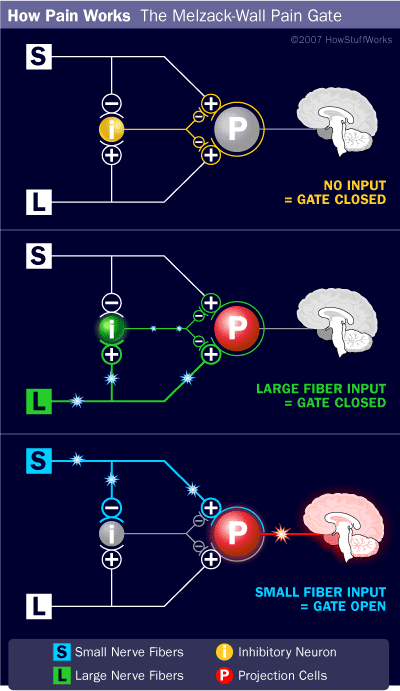
- When no input comes in, the inhibitory neuron prevents the projection neuron from sending signals to the brain (gate is closed).
- Normal somatosensory input happens when there is more large-fiber stimulation (or only large-fiber stimulation). Both the inhibitory neuron and the projection neuron are stimulated, but the inhibitory neuron prevents the projection neuron from sending signals to the brain (gate is closed).
- Nociception (pain reception) happens when there is more small-fiber stimulation or only small-fiber stimulation. This inactivates the inhibitory neuron, and the projection neuron sends signals to the brain informing it of pain (gate is open).
Descending pathways from the brain close the gate by inhibiting the projector neurons and diminishing pain perception.
This theory doesn’t tell us everything about pain perception, but it does explain some things. If you rub or shake your hand after you bang your finger, you stimulate normal somatosensory input to the projector neurons. This closes the gate and reduces the perception of pain.
What is The Gate Control Theory ?
Most, if not all, ailments of the body cause pain. Pain is interpreted and perceived in the brain. Pain is modulated by two primary types of drugs that work on the brain: analgesics and anesthetics. The term analgesic refers to a drug that relieves pain without loss of consciousness. The term central anesthesia refers to a drug that depresses the CNS. It is characterized by the absence of all perception of sensory modalities, including loss of consciousness without loss of vital functions.
Opiate Analgesia (OA)
The most effective clinically used drugs for producing temporary analgesia and relief from pain are the opioid family, which includes morphine, and heroin. There are currently no other effective pain therapeutic alternatives to opiates. Several side effects resulting from opiate use include tolerance and drug dependence (addiction). In general, these drugs modulate the incoming pain information in the spinal and central sites, as well as relieve pain temporarily, and are also known as opiate producing analgesia(OA). Opiate antagonist is a drug that antagonizes the opioid effects, such as naloxone or maltroxone, etc. They are competitive antagonists of opiate receptors. The brain has a neuronal circuit and endogenous substances to modulate pain.
Endogenous Opioids
Opioidergic neurotransmission is found throughout the brain and spinal cord and appears to influence many CNS functions, including nociception, cardiovascular functions, thermoregulation, respiration, neuroendocrine functions, neuroimmune functions, food intake, sexual activity, aggressive locomotor behavior as well as learning and memory. Opioids exert marked effects on mood and motivation and produce euphoria.
Three classes of opioid receptors have been identified: μ-mu, δ-delta and κ-kappa. All three classes are widely distributed in the brain. The genes encoding each one of them have been cloned and found to be members of the G protein receptors. Moreover, three major classes of endogenous opioid peptides that interact with the above opiate receptors have been recognized in the CNS:β-endorphins, enkephalins and the dynorphins. These three opioid peptides are derived from a large protein precursor by three different genes: the proopiomelanocortin (POMC) gene, the proenkephalin gene and the prodynorphin gene. The opioid peptides modulate nociceptive input in two ways: 1) block neurotransmitter release by inhibiting Ca2+ influx into the presynaptic terminal, or 2) open potassium channels, which hyperpolarizes neurons and inhibits spike activity. They act on various receptors in the brain and spinal cord. Enkephalins are considered the putative ligands for the δ receptors, β endorphins for the μ-receptors, and dynorphins for the κ receptors. The various types of opioid receptors are distributed differently within the central and peripheral nervous system. There is evidence for functional differences in these receptors in various structures. This explains why many unwanted side effects occur following opiate treatments. For example, mu (μ) receptors are widespread in the brain stem parabrachial nuclei, a respiratory center and inhibition of these neurons elicits respiratory depression.
Central or peripheral terminals of nociceptive afferent fibers contain opiate receptors where exogenous and endogenous opioids could act to modulate the ability to transmit nociceptive information. Moreover, high densities of opiate receptors are found in periaqueductal gray (PAG), nucleus raphe magnus (NRM), and dorsal raphe (DR) in the rostral ventral medulla, in the spinal cord, caudate nucleus (CN), septal nucleus, hypothalamus, habenula and hippocampus. Additional detailson opiate receptors is provided later in this lecture.
Systemically administered opioids at analgesic doses activate spinal and supraspinal mechanisms via μ, δ, and κ type opioid receptors and modulate pain signals.
Gate Control theory
The first pain modulatory mechanism called the “Gate Control” theory was proposed by Melzack and Wall in the mid 1960s. The concept of the gate control theory is that non-painful input closes the gates to painful input, which results in prevention of the pain sensation from traveling to the CNS (i.e., non-noxious input [stimulation] suppresses pain).
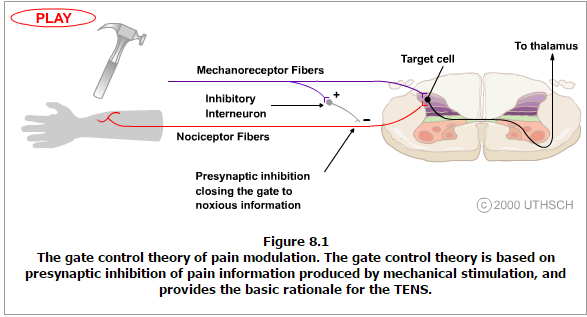
The theory suggests that collaterals of the large sensory fibers carrying cutaneous sensory input activate inhibitory interneurons, which inhibit (modulate) pain transmission information carried by the pain fibers. Non-noxious input suppresses pain, or sensory input “closes the gate” to noxious input (Figure 8.1). The gate theory predicts that at the spinal cord level, non-noxious stimulation will produce presynaptic inhibition on dorsal root nociceptor fibers that synapse on nociceptors spinal neurons (T), and this presynaptic inhibition will block incoming noxious information from reaching the CNS (i.e., will close the gate to incoming noxious information).
The gate theory was the rationale for the idea behind the production and the use of transcutaneous electrical nerve stimulation (TENS) for pain relief. To be effective, the TENS unit produces two different current frequencies below the pain threshold that can be tolerated by the patient. This procedure has partial success in pain therapy.
Stimulation produced analgesia (SPA)
Evidence for an intrinsic analgesia system was demonstrated by intracranial electrical stimulation of certain discrete brain sites. These areas are the periaqueductal gray (PAG) and nucleus raphe magnus (NRM), dorsal raphe (DR), caudate nucleus (CN), septal nucleus (Spt) and other nuclei. Such stimulation inhibits pain, (i.e., producing analgesia without behavioral suppression), while the touch, pressure and temperature sensation remain intact. SPA is more pronounced and lasts a longer time after stimulation in humans than in experimental animals. Moreover, during SPA, the subjects still respond to nonpainful stimuli such as touch and temperature within the circumscribed area of analgesia. The most effective CNS sites for SPA are the PAG and the raphe nuclei (RN).
Electrical stimulation of PAG or NRM inhibits spinal thalamic cells, (i.e. spinal neurons that project monosynaptically to the thalamus) in laminae I, II and V so that the noxious information from the nociceptors are modulated at the spinal cord level. PAG has neuronal connections to NRM.
The action of the PAG most likely occurs by activation of the descending pathway from NRM and probably also by activation of ascending connections acting on higher subcortical levels of the CNS. Moreover, electrical stimulation of PAG or NRM produces behavioral analgesia, (i.e., stimulation produced analgesia, see Figure 8.2). Stimulation produced analgesia (SPA) elicits release of endorphin and is blocked by the opiate antagonist naloxone.
During PAG and/or RN stimulation, serotonin (5-HT) is also released from ascending and descending axons in subcortical nuclei, in spinal trigeminal nuclei and in the spinal cord. This release of 5-HT modulates pain transmission by inhibiting incoming sensory activity. Depletion of 5-HT by electrical lesion of the raphe nuclei or by a neurotoxic lesion produced by local injection of a chemical agent like parachlorophenylalanine (PCPA) results in blocking the ability of both opiate (intracranial and systemic) and electrical stimulation to produce analgesia.
To verify whether the electrical stimulation produced analgesia via the release of opiate and serotonin, the area was locally microinjected with morphine or 5-HT. These microinjections indeed produce analgesia (Figure 8.3). These procedures also provide a method of identifying brain regions associated with pain suppression and help to produce a map of pain centers. The most effective method of producing opiate analgesia (OA) is by intracerebral injection of morphine into the PAG.
The PAG and RN and other brain structures where analgesia is produced are also rich in opiate receptors. Intracerebral opioid administration produced analgesia and SPA can be blocked by either systemic or by local microinjections of naloxone, the morphine antagonist, into the PAG or RN. Therefore, it has been suggested that the two (OA and SPA) operate by a common mechanism.
If OA and SPA act through the same intrinsic system, then the hypothesis that opiates activate a pain-suppression mechanism is more likely. In fact, present evidence indicates that microinjections of an opiate into the PAG activate an efferent brainstem system that suppresses pain transmission at segmental (spinal cord) levels. These observations indicate that analgesia elicited from the PAG requires a descending pathway to the spinal cord.
Gate_Control_Theory_of_Pain
More information about Gate Control Theory, Please check here.
Pain Medications, Pain Relief, and Pain Management