What is Gabapentin and What is Gabapentin Used for ?
Gabapentin is used with other medications to prevent and control seizures. It is also used to relieve nerve pain following shingles (a painful rash due to herpes zoster infection) in adults. Gabapentin is known as an anticonvulsant or antiepileptic drug.
OTHER USES: This section contains uses of this drug that are not listed in the approved professional labeling for the drug but that may be prescribed by your health care professional. Use this drug for a condition that is listed in this section only if it has been so prescribed by your health care professional.
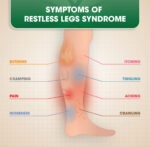
Gabapentin may also be used to treat other nerve pain conditions (such as diabetic neuropathy, peripheral neuropathy, trigeminal neuralgia) and restless legs syndrome.
Gabapentin is an anti-epileptic medication, also called an anticonvulsant. It affects chemicals and nerves in the body that are involved in the cause of seizures and some types of pain.
Gabapentin is used in adults to treat nerve pain caused by herpes virus or shingles (herpes zoster).
The Horizant brand is also used to treat restless legs syndrome (RLS).
The Neurontin brand is also used to treat seizures in adults and children who are at least 3 years old.
Use only the brand and form of gabapentin that your doctor has prescribed. Check your medicine each time you get a refill at the pharmacy, to make sure you have received the correct form of this medication.
Gabapentin is also commonly prescribed for many off-label uses, such as treatment of restless leg syndrome, anxiety disorders, insomnia, and bipolar disorder. There are, however, concerns regarding the quality of the trials conducted and evidence for some such uses, especially in the case of its use as a mood stabilizer in bipolar disorder.

What is your Experience with Gabapentin?

This is a good question, in that it made me revisit this topic after a long time and gave me some new perspectives.
Firstly, some general comments:
- FDA approval of any drug for any indication is always subject to updation
- Non-approved usage of a medicine happens all the time; the only legal binding is that a company cannot promote/market an off-label use for a drug (this is my own conclusion, subject to correction)
- FDA approval is not binding on countries other than the US
Now for some specific comments (I am quoting directly from this article: Gabapentin Therapy in Psychiatric Disorders: A Systematic Review, by Berlin, Butler, and Perloff (the points are so good, I didn’t bother editing or paraphrasing, except for some bolding and ellipsis)
- Gabapentin was originally approved by the US Food and Drug Administration (FDA) for the treatment of partial seizures in 1993
- Subsequent approval for postherpetic neuralgia in 2002
- Within a decade of initial FDA approval, gabapentin’s second most common use became off-label prescription for psychiatric disorders (mainly for anxiety disorders)
- Gabapentin’s use in psychiatric disorders has been shrouded in controversy, from the 1996 lawsuit against Warner-Lambert for promoting Neurontin for off-label indications, including psychiatric disorders
- Gabapentin has a limited, generally well-tolerated side effect profile, and … has minimal drug-drug interactions
- Gabapentin has … a proposed mechanism well-established for treating neuropathic pain and seizure
- Numerous case reports and reviews suggest gabapentin’s potential efficacy as either monotherapy or adjunctive therapy in the treatment of bipolar disorder, depression, anxiety disorders, posttraumatic stress disorder (PTSD), alcohol dependence, and other types of drug abuse.
The authors then did a very thorough search for articles on studies, case reports or reviews looking into the (off-label) use of Gabapentin for various psychiatric conditions.
They conclude that:
- Gabapentin does appear to provide benefit for some anxiety disorders, although randomized controlled trials have been limited to social phobia, anxiety in breast cancer, and perioperative anxiety.
- To date, no studies exist for gabapentin efficacy in generalized anxiety disorder.
- There is limited evidence to suggest the use of gabapentin in depression, PTSD, and OCD
- Multiple studies suggest gabapentin has some efficacy in alcohol dependence, withdrawal, and craving.
- As for gabapentin’s use in other types of substance dependence, there are no data to support its efficacy in cocaine or methamphetamine dependence.
Here is an application to the “21st meeting of the WHO Expert Committee on Selection and Use of Essential Medicines” for the inclusion of gabapentin on the WHO Model List of Essential Medicines, submitted by International Association for the Study of Pain (IASP), the Neuropathic Pain Special Interest Group (NeuPSIG) of the IASP, and the International Association of Hospice and Palliative Care (IAHPC), and supported by similar bodies in numerous other countries.
Here is the opening statement from this application:
We are applying for the inclusion of gabapentin as an analgesic agent for the management of neuropathic pain (central and peripheral) in adults. The medicine has regulatory approval for the treatment of several neuropathic pain states in adults by numerous stringent regulatory bodies (including the Food and Drug Administration [1] and European Medicines Agency [2]).
I am not aware of the outcome of this application (haven’t had the time to search for the relevant information).
To balance it all, here is a very good article (by Goodman & Brett): Gabapentin and Pregabalin for Pain — Is Increased Prescribing a Cause for Concern?
taking a very critical look at all the off-label use of these medicines, the paucity of evidence, and the poor quality of research that exists in this particular area.
Finally, some personal comments from me:
- Lack of evidence does not mean there is evidence of lack (of efficacy, for example)
- Clinical Trials, including RCTs are good evidence, but still not absolute
- Off-label use for any drug is between the doctor and the patient, with mutual understanding and informed consent
- The use of Gabapentin as a sedative is neither well-studied, nor recommended.
- Mild to moderate sedation is a common enough side-effect for patients and doctors to be tempted to use it that way; but if you have no other use for Gabapentin, then you are better off not taking it – there are many other better drugs for that.