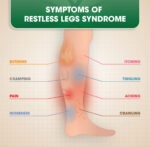
Gabapentin is a medication that belongs to the class of drugs known as anticonvulsants or antiepileptics. It is primarily used to treat seizures and neuropathic pain, but it is also used off-label for various other conditions such as anxiety, insomnia, and restless leg syndrome.
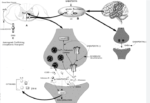
Gabapentin works by binding to a specific type of voltage-gated calcium channel (the alpha-2-delta subunit) in the brain and spinal cord. This reduces the release of various neurotransmitters, including glutamate, substance P, and noradrenaline, which are involved in pain perception and seizures.
Gabapentin is rapidly absorbed after oral administration, and its bioavailability is not affected by food. The peak plasma concentration is reached within 2-3 hours after administration. Gabapentin is not metabolized in the liver and is primarily eliminated by renal excretion.
The half-life of gabapentin is approximately 5-7 hours, and it is typically administered three times a day to maintain therapeutic levels in the blood. The dosage of gabapentin may need to be adjusted in patients with renal impairment, and it should be used with caution in patients with a history of substance abuse or suicidal ideation.
Overall, gabapentin is a well-tolerated medication, but common side effects may include dizziness, drowsiness, fatigue, and ataxia. Gabapentin may also increase the risk of suicidal thoughts or behaviors, particularly in younger patients. Therefore, close monitoring is recommended during treatment with gabapentin.
In 2009 the U.S. Food and Drug Administration issued a warning of an increased risk of depression and suicidal thoughts and behaviors in patients taking gabapentin, along with other anticonvulsant drugs modifying the packaging insert to reflect this. In July 2009 the manufacturer of gabapentin (Pfizer) went to trial regarding the association between gabapentin and the increased risk of suicide.
Antiepileptic drugs (AEDs), including Neurontin, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.

The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5-100 years) in the clinical trials analyzed. Table 2 shows absolute and relative risk by indication for all evaluated AEDs.
Table 1 : Risk by indication for antiepileptic drugs in the pooled analysis
| PLACEBO PATIENTS WITH EVENTS PER 1000 PATIENTS | DRUG PATIENTS WITH EVENTS PER 1000 PATIENTS | RELATIVE RISK: INCIDENCE OF EVENTS IN DRUG PATIENTS/INCIDENCE IN PLACEBO PATIENTS | RISK DIFFERENCE: ADDITIONAL DRUG PATIENTS WITH EVENTS PER 1000 PATIENTS | |
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing Neurontin or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
Pain Medications, Pain Relief, and Pain Management