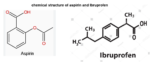
Ibuprofen was initially introduced as a prescription NSAID used for such conditions as rheumatoid arthritis, in 1969 in the UK and 1974 in the US. Ibuprofen was the first member of Propionic acid derivatives introduced in 1969. It is a popular domestic and over the counter analgesic and antipyretic for adults and children. Ibuprofen has been rated as the safest conventional NSAID by spontaneous adverse drug reaction reporting systems in the UK. This article summarizes the main pharmacological effects, therapeutical applications and adverse drug reactions, drug-drug interactions and food drug interactions of ibuprofen that have been reported especially during the last 10 years.
Ibuprofen’s safety record remained good with problems only being related to overdosing. The product was “Fast-Tracked” from prescription to OTC (over-the-counter) status by the FDA.
Ibuprofen is (2RS)-1[4-(2-methyl propyl) phenyl] propionic acid (BP. 2004). Ibuprofen was the first member of propionic acid derivatives to be introduced in 1969 as a better alternative to Aspirin. Gastric discomfort, nausea and vomiting, though less than aspirin or indomethacin, are still the most common side effects.
Ibuprofen is the most commonly used and most frequently prescribed NSAID. It is a non-selective inhibitor of cyclo-oxygenase-1 (COX-1) and Cyclooxygenase-2 (COX-2).4 Although its anti inflammatory properties may be weaker than those of some other NSAIDs, it has a prominent analgesic and antipyretic role. Its effects are due to the inhibitory actions on cyclo-oxygenases, which are involved in the synthesis of prostaglandins. Prostaglandins have an important role in the production of pain, inflammation and fever.
It became available in the UK in 1983 under the brand name “Nurofen” and in the United States in 1984 under the brand name “Advil”.
Anyone could buy it without a prescription for the treatment of headaches, dental pain, migraine and menstrual pain.
The first automated production facility opened in Hammonton, New Jersey in 1988. The design of the tablets 67 layer structure came from George Van Parys and Webb Crew both who worked for American Home Products at the time.
It was soon recognized that output capacity was insufficient, and production was inefficient. A new facility was built in Guayama, Puerto Rico, which comprised an initial 20 new systems. Van Parys created the systems and built the plant. Each system now produced 540,000 tablets every 9 hours, or 1,440,000 tablets per day per system. This gave Whitehall the ability to produce a total of 876,600,000 tablets per month.
Whitehall-Robbins closed the Hammonton manufacturing facility in 1996, moving its 10 production units to Rouses Point, New York. This move was short lived, and the units ended up in Guayama in 2004. The units were eventually moved to China to cut the labor cost.
Pain Medications, Pain Relief, and Pain Management